Case Description
A 51-year-old right-handed woman reported left trigeminal (V3) distribution lancinating pain gradually progressive over 24 months. Triggers included chewing, talking, brushing her teeth, drinking or moving her mouth. She had some relief with carbamazepine and gabapentin but did not tolerate the medications well and was transitioned to oxcarbazepine. The pain subsequently recurred with increasing intensity refractory to increased doses of oxcarbazepine. Neurological examination was normal with no evidence of trigeminal sensory or motor deficits.
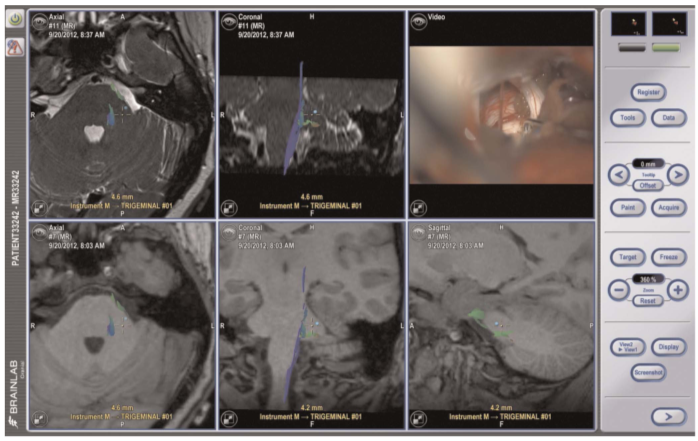
There was no arterial compression of the left trigeminal nerve shown on a brain MRI with diffusion tensor imaging (DTI) sequences. The decision was made to take her to surgery for a left-sided microvascular decompression (MVD) and possible tractotomy via a retrosigmoid approach. At surgery, a compressing vein over the dorsum of the left trigeminal nerve was noted and dissected off the nerve; a muscle graft was gently wedged in place to separate the vein from the nerve. No convincing arterial compression was found. A tractotomy of the descending nucleus of the trigeminal nerve was performed using stereotactic neuronavigation and microscopic visualization of the inferior aspect of the trigeminal nerve root entry zone; the preferred entry point for the VPT procedure is 1mm inferolateral to this point (Figure 1).
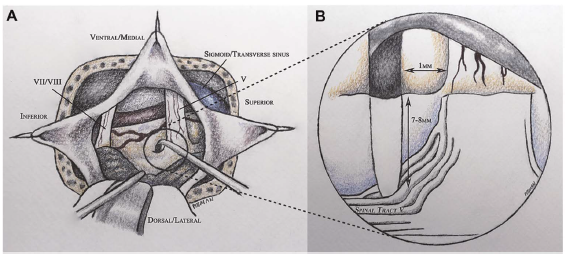
Low power micro-bipolar electrocautery is used to incise the pia and a 6-8 mm right-angled microdissector (tracked by the neuronavigation system) is used to enter into the brainstem to a depth of 7-9 mm in a plane parallel to the trigeminal nerve proceeding dorsomedially to the descending spinal tract of the trigeminal nerve (Figure 1). Neuronavigation is used to guide the microdissector 2-3 mm along the inferior aspect of the pontine descending trigeminal pathway. This action facilitates the disruption of the spinal tract of the trigeminal nerve superior to the point at which it converges into a vertical compact tract (Figures 1 and 2). The anterolateral corner of the rhomboid fossa and fourth ventricular floor plane is used to guide the trajectory of the microdissector (Figure 2).
The patient made an uneventful and excellent postoperative recovery. She reported resolution of her left sided facial pain immediately following surgery but remained on a low dose of oxcarbazepine for persistent jaw discomfort. At her last follow-up, four months after surgery, she reported she was doing well with no pain, allowing for weaning and complete cessation of her oxcarbazepine.
Discussion
Trigeminal neuralgia (TN) is a pain syndrome that is characterized by paroxysmal brief episodes of lancinating pain in one or more divisions of the trigeminal nerve classically triggered by chewing, speaking, swallowing, touching the face or brushing the teeth. It is usually unilateral and can have a dramatic effect on normal daily activities with resultant deleterious effects on hygiene, nutrition and the overall psychological health of patients. Pain-free intervals are also common, distinguishing classic TN from other facial pain syndromes that may mimic it. However, recurrences or relapses of pain are also common and are usually experienced in the same distribution as the initial pain (1).
TN can be categorized into seven different classifications based on etiology:
- TN type 1 (TN1): idiopathic with predominantly lancinating paroxysmal pain;
- TN type 2 (TN2): idiopathic with background dysesthetic pain;
- TN3: unintentional trigeminal injury secondary to facial trauma;
- TN4: a deafferentation pain syndrome that includes patients with TN secondary to intentional procedures to the trigeminal nerve;
- TN5: trigeminal pain associated with multiple sclerosis;
- TN6: post-herpetic TN associated with facial herpes zoster; or
- TN7: includes patients who have somatoform pain disorder with psychological evaluation (2).
Durable pain relief with minimal untoward effects is the goal when treating trigeminal neuralgia. Medical therapy may be used initially (primarily carbamazepine or oxcarbazepine), but surgical intervention is effective treatment when neurovascular compression (NVC) is present. Most practicing neurosurgeons agree that relief is best achieved with microvascular decompression (MVD) if there is NVC of the trigeminal nerve (first proposed as a cause of TN by Dandy in 1934). Magnetic resonance imaging (MRI) 3-D fast imaging using steady state acquisition (FIESTA) sequences accurately reveal NVC at the root entry zone (REZ) and can be used to predict which patients may benefit from MVD. However, intraoperative NVC can be less impressive than suggested by MRI FIESTA prior to surgery; often a vessel is contacting cranial nerve V without compressing or distorting it (4). Additionally, TN may also exist in the absence of NVC (3). Although less common than NVC, these include tumors, multiple sclerosis (MS), root injury or demyelination disorders. The surgical treatment for these patients is different. Given the absence of NVC, alternatives to MVD in these patients include internal neurolysis, radiofrequency ablation, radiosurgery, glycerol injections, partial sensory rhizotomy, balloon rhizotomy and tractotomy of the descending spinal tract of the trigeminal nerve (1,4-6). (Discussed separately here)
Management of TN begins with optimizing medical management of the patient’s symptoms. If the pain becomes refractory to medical management or if the patient is unable to tolerate medical management secondary to side effects, surgical interventions are discussed. Based on MRI imaging, MVD alone, VPT alone or a combined procedure may be recommended; only those patients without remarkable arterial NVC receive VPT. MVD may be performed in conjunction with VPT, even when NVC is present, if the source of compression is a venous structure or the arterial compression is less convincing under the microscope at surgery.
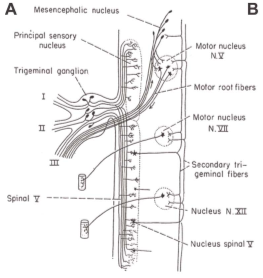
VPT is a modification of various other tractotomy procedures, including that of the descending tract of the trigeminal nerve, first introduced by Sjöqvist in 1938 (7-13), largely abandoned due to complications, including lower cranial nerve dysfunction, proprioceptive issues and dysesthesias. The technique employed by the VPT targets the fibers of the spinal trigeminal tract (SpTV) before they coalesce into a compact tract (Figure 3). In this initial portion, the fibers of the SpTV form a lenticular shaped elbow after they separate from the intrapontine portion of the trigeminal nerve. This elbow is just ventrolateral to the principal sensory and motor trigeminal nuclei. By using this as a target, the tractotomy mechanically disrupts the temperature and pain fibers from the face and stops short of the spinal nucleus or motor nucleus of the trigeminal nerve. The VPT also disrupts fibers cephalad to where the sensory fibers of VII, IX or X join the SpTV, therefore avoiding damage to those fibers. This method was devised in order to minimize the risk of other complications, such as anesthesia dolorosa or neurotrophic keratitis that can occur after more generalized damage to the trigeminal nerve after rhizotomy or radiofrequency ablation.
Our series of TN surgical patients include 72 (35 percent) patients who have undergone VPT, whether as a stand-alone procedure or in combination with MVD. Recurrent pain at an average of 22 months was seen in 13 patients (18 percent) with nine of these patients achieving pain controlled with medication postoperatively (Barrow Neurological Institute (BNI) Pain Intensity Score score IIIa).
As with MVD, there are always the surgical risks associated with invasive procedures of infection and spinal fluid leaks, along with postoperative facial numbness and weakness. VPT carries the additional risk of missing the descending spinal tract of the trigeminal nerve due to normal anatomic variation and possibly destroying adjacent sensory and motor fibers. In one patient, concern about neuronavigation accuracy prevented VPT. Overall, this has been a very well tolerated and successful procedure and none of the patients have experienced lasting iatrogenic neurological deficits following VPT.
THE EXPERTS WEIGH IN
Sepehr Sani, MD, faans, chicago
 The authors present the surgical technique which involves the lesioning of the descending spinal trigeminal tract fibers in a series of 79 patients undergoing MVD for TN via a retrosigmoid craniotomy. They report adequate pain relief in an impressive 94 percent of patients at an average follow up of 22 months. While this approach seems safe and effective in instances where no significant neurovascular compression is found during surgery, I typically prefer starting with less invasive approaches in lieu of vascular compression seen on preoperative MRI. I offer percutaneous radiofrequency ablation to patients that present with severe, medication refractory TN without evidence of vascular compression on MRI as this therapy has been shown to provide initial pain relief that is similar to MVD (14,15). Although the durability of pain relief is inferior to that of MVD, it is less invasive and can be repeated in cases of recurrence (15).
The authors present the surgical technique which involves the lesioning of the descending spinal trigeminal tract fibers in a series of 79 patients undergoing MVD for TN via a retrosigmoid craniotomy. They report adequate pain relief in an impressive 94 percent of patients at an average follow up of 22 months. While this approach seems safe and effective in instances where no significant neurovascular compression is found during surgery, I typically prefer starting with less invasive approaches in lieu of vascular compression seen on preoperative MRI. I offer percutaneous radiofrequency ablation to patients that present with severe, medication refractory TN without evidence of vascular compression on MRI as this therapy has been shown to provide initial pain relief that is similar to MVD (14,15). Although the durability of pain relief is inferior to that of MVD, it is less invasive and can be repeated in cases of recurrence (15).
Stereotactic radiosurgery (SRS) is another even less invasive option that can be considered in patients without clear neurovascular compression on MRI. While less effective than percutaneous radiofrequency ablation and MVD, several studies have demonstrated satisfactory pain relief between 2.5-12 months but with less durable long-term relief (16-18). Because pain relief is usually not immediate, I typically recommend this option for patients whose symptoms are less severe or wish to forego invasive options. Generally, when these less invasive options have failed in patients without clear neurovascular compression on MRI, I will offer exploratory MVD with neurolysis.
The incorporation of diffusion tensor imaging tractography and stereotactic navigation software is a welcome adjunct to the ventral pontine tractotomy (VPT) procedure which itself was described eighty years ago by Sjöqvist. However, significant complications including destruction of trigeminal sensory and/or motor fibers can arise if the accuracy of the stereotactic software is distorted by factors such as brainstem shift. Henceforth, safe and successful outcomes depend on the surgeon’s experience with the anatomy and variations herein. Thus, while the complication rate may be low with impressive outcomes in the hands of highly experienced surgeons as above, such results may not be generalizable.
While VPT appears to be effective in most patients with TN, there still remains a subset of patients that remain refractory to treatment, suggesting at least partly a central component in its pathophysiology. Recent work by Hung et al. has explored the use of preoperative DTI metrics to determine patient specific response to peripheral treatment (19). They found that changes within the pontine segment of the nerve distinguished responders from non-responders thus indicating that central changes within the brainstem, possibly induced from longstanding peripheral insults such as neurovascular compression, may be responsible for lack of treatment response. Further treatment options aimed at treating the central pain component may provide more efficacious and durable pain relief to patients with treatment refractory TN. As we gain a better understanding of the pathophysiology of TN, improvements in preoperative imaging modalities may help to better guide the appropriate initial therapy for patients.
7. Sjöqvist, O. (1938). Vi. Section of the bulbospinal fifth tract for the relief of facial pain. Acta Psychiatrica Scandinavica, 13(S17), 93-95.
8. White, J. C., & Sweet, W. (1969). Pain and the neurosurgeon: A forty-year experience. 1000.
10. Kunc, Z. & Simek, J. (1955). Glossopharyngeal neuralgia cured by tractotomy according to Sjöqvist [In Czech]. Neurologie a Psychiatrie Ceskoslovenská, 18(2),135-137.
[aans_authors]